|
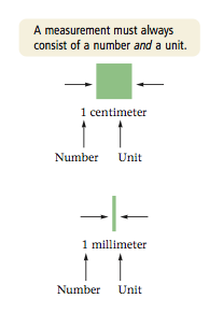
A quantitative observation is called a measurement. A measurement always consist of two parts: a number and a unit. Both parts are necessary to make the measure measurement meaningful.
Sections 2.1 Scientific Notation 2.2 Units 2.3 Measurements of Length, Volume, and Mass 2.4 Uncertainty in Measurement 2.5 Significant Figures 2.6 Problem Solving and Dimensional Analysis 2.7 Temperature Conversions: An Approach to Problem Solving 2.8 Density |
- Home
- Chapters Part 1
- Chapter 1
- Chapter 2>
- Chapter 3>
- Chapter 4>
- Chapter 11>
- 11.1 Rutherford's Atom
- 11.2 Electromagnetic Radiation
- 11.3 Emission of Energy by Atom
- 11.4 The Energy Levels of Hydrogen
- 11.5 The Bohr Model of the Atom
- 11.6 The Wave Mechanical Model of the Atom
- 11.7 The Hydrogen Orbitals
- 11.8 The Wave Mechanical Model: Further Development
- 11.9 Electron Arrangements in the First Eighteen Atoms on the Periodic Table
- 11.10 Electron Configuration and the Periodic Table
- 11.11 Atomic Properties and the Periodic Table
- Chapter 12>
- 12.1 Types of Chemical Bonds
- 12.2 Electronegativity
- 12.3 Bond Polarity and Dipole Moments
- 12.4 Stable Electron Configurations and Charges on Ions
- 12.5 Ionic Bonding and Structures of Ionic Compounds
- 12.6 Lewis Structures
- 12.7 Lewis Structures of Molecules with Multiple Bonds
- 12.8 Molecular Structure
- 12.9 Molecular Structure: The VSEPR Model
- 12.10 Molecular Structure: Molecules with Double Bonds
- Chapter 5>
- Chapter 6>
- Chapter 7>
- Chapters Part 2