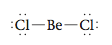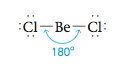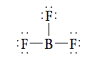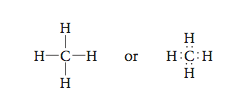OBJECTIVE: To learn to predict molecular geometry from the number of electron pairs.
Many experimental methods now exist for determining the molecular structure of a molecule—that is, the three-dimensional arrangement of the atoms. These methods must be used when accurate information about the structure is required. However, it is often useful to be able to predict the approximate molecular structure of a molecule. In this section we consider a simple model that allows us to do this. This model, called the valence shell electron pair repulsion (VSEPR) model, is useful for predicting the molecular structures of molecules formed from nonmetals. The main idea of this model is that the structure around a given atom is determined by minimizing repulsions between electron pairs. This means that the bonding and nonbonding electron pairs (lone pairs) around a given atom are positioned as far apart as possible.
To see how this model works, we will first consider the molecule BeCl2, which has the following Lewis structure (it is an exception to the octet rule):
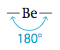
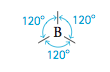
Note that there are two pairs of electrons around the beryllium atom. What arrangement of these electron pairs allows them to be as far apart as possible to minimize the repulsions? The best arrangement places the pairs on opposite sides of the beryllium atom at 180° from each other.
This is the maximum possible separation for two electron pairs. Now that we have determined the optimal arrangement of the electron pairs around the central atom, we can specify the molecular structure of BeCl2—that is, the positions of the atoms. Because each electron pair on beryllium is shared with a chlorine atom, the molecule has a linear structure with a 180° bond angle.
Whenever two pairs of electrons are present around an atom, they should always be placed at an angle of 180 to each other to give a linear arrangement.
Next let’s consider BF3, which has the following Lewis structure (it is an- other exception to the octet rule):
Next let’s consider BF3, which has the following Lewis structure (it is an- other exception to the octet rule):
|
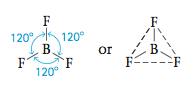
Here the boron atom is surrounded by three pairs of electrons. What arrangement minimizes the repulsions among three pairs of electrons? Here the greatest distance between electron pairs is achieved by angles of 120°.
Because each of the electron pairs is shared with a fluorine atom, the molecular structure is
This is a planar (flat) molecule with a triangular arrangement of F atoms, commonly described as a trigonal planar structure. Whenever three pairs of electrons are present around an atom, they should always be placed at the corners of a triangle (in a plane at angles of 120° to each other).
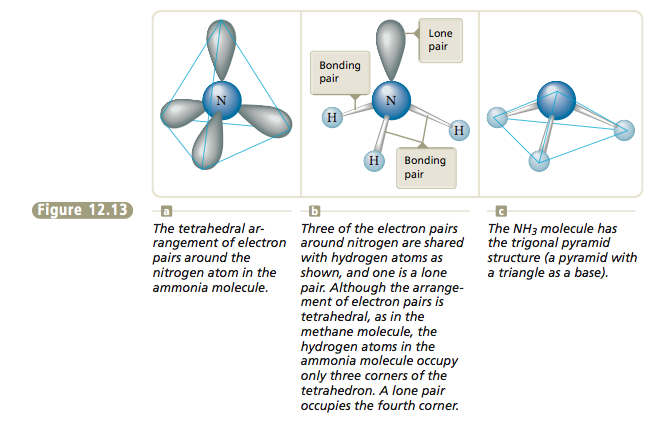
Next let’s consider the methane molecule, which has the Lewis structure The carbon atom and the electron pairs are all in a plane represented by the surface of the paper, and the angles between the pairs are all 90°.
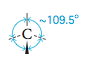
Is there another arrangement with angles greater than 90° that would put the electron pairs even farther away from each other? The answer is yes. We can get larger angles than 90° by using the following three-dimensional structure, which has angles of approximately 109.5°. |
|
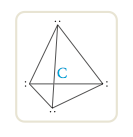
In this drawing the wedge indicates a position above the surface of the paper and the dashed lines indicate positions behind that surface. The solid line indicates a position on the surface of the page. The figure formed by connecting the lines is called a tetrahedron, so we call this arrangement of electron pairs the tetrahedral arrangement.
This is the maximum possible separation of four pairs around a given atom.
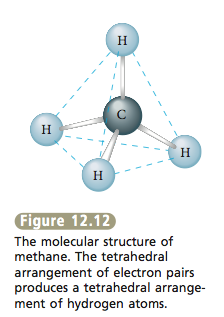
Whenever four pairs of electrons are present around an atom, they should always be placed at the corners of a tetrahedron (the tetrahedral arrangement). Now that we have the arrangement of electron pairs that gives the least repulsion, we can determine the positions of the atoms and thus the mo- lecular structure of CH4. In methane each of the four electron pairs is shared between the carbon atom and a hydrogen atom. Thus the hydrogen atoms are placed as shown in Figure 12.12, and the molecule has a tetrahedral structure with the carbon atom at the center. |
|
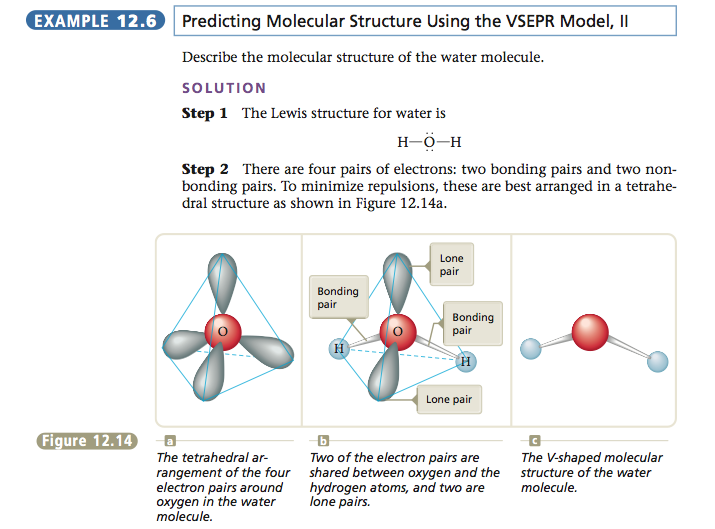
Recall that the main idea of the VSEPR model is to find the arrangement of electron pairs around the central atom that minimizes the repulsions. Then we can determine the molecular structure by knowing how the electron pairs are shared with the peripheral atoms. A systematic procedure for using the VSEPR model to predict the structure of a molecule is outlined below.
|
The various molecules we have considered are summarized in Table 12.4 on the following page. Note the following general rules.