OBJECTIVE: To understand how the emission spectrum of hydrogen demonstrates the quantized nature of energy.
|
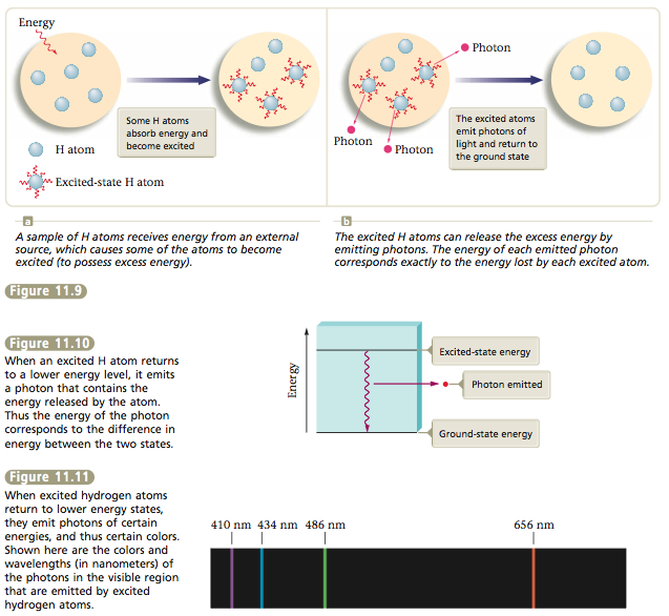
As we learned in the last section, an atom with excess energy is said to be in an excited state. An excited atom can release some or all of its excess energy by emitting a photon (a “particle” of electromagnetic radiation) and thus move to a lower energy state. The lowest possible energy state of an atom is called its ground state.
We can learn a great deal about the energy states of hydrogen atoms by observing the photons they emit. To understand the significance of this, you need to remember that the different wavelengths of light carry different amounts of energy per photon. When a hydrogen atom absorbs energy from some outside source, it uses this energy to enter an excited state. It can release this excess energy (go back to a lower state) by emitting a photon of light (Figure 11.9). We can picture this process in terms of the energy-level diagram shown in Figure 11.10. The important point here is that the energy contained in the photon corresponds to the change in energy that the atom experiences in going from the excited state to the lower state. |
|
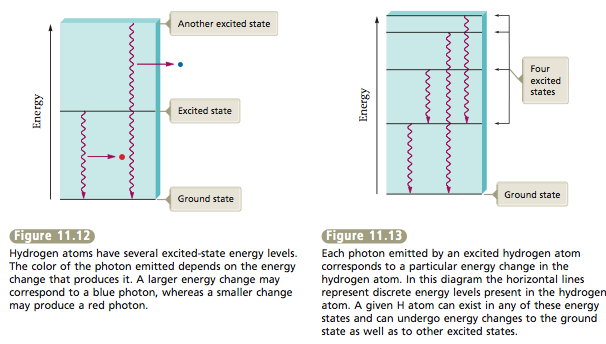
Because only certain photons are emitted, we know that only certain energy changes are occurring (Figure 11.12). This means that the hydrogen atom must have certain discrete energy levels (Figure 11.13). Excited hydrogen atoms always emit photons with the same discrete colors (wavelengths)— those shown in Figure 11.11. They never emit photons with energies (colors) in between those shown. So we can conclude that all hydrogen atoms have the same set of discrete energy levels. We say the energy levels of hydrogen are quantized. That is, only certain values are allowed. Scientists have found that the energy levels of all atoms are quantized.
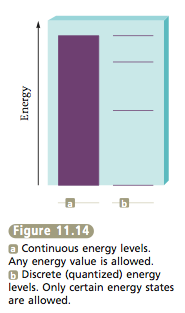
The quantized nature of the energy levels in atoms was a surprise when scientists discovered it. It had been assumed previously that an atom could exist at any energy level. That is, everyone had assumed that atoms could have a continuous set of energy levels rather than only certain discrete values (Figure 11.14). A useful analogy here is the contrast between the eleva- tions allowed by a ramp, which vary continuously, and those allowed by a set of steps, which are discrete (Figure 11.15). The discovery of the quantized nature of energy has radically changed our view of the atom, as we will see in the next few sections. |
Extra Link
You can also visit this link. It could help improve your understanding on the energy level of hydrogen..
You can also visit this link. It could help improve your understanding on the energy level of hydrogen..