|
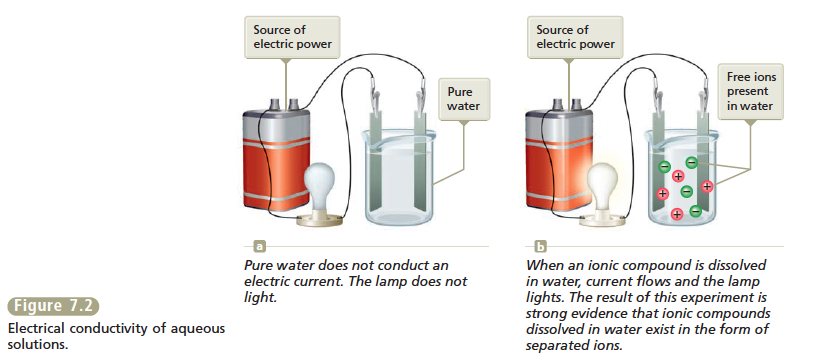
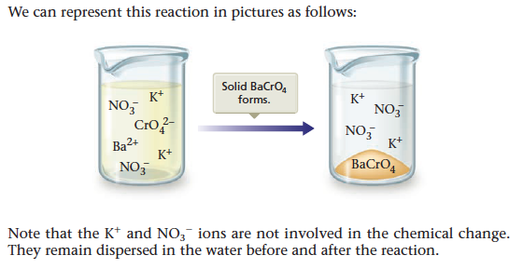
One driving force for a chemical reaction is the formation of a solid, a process called precipitation. The solid that forms is called a precipitate, and the reaction is known as a precipitation reaction. For example, when an aqueous (water) solution of potassium chromate, K2CrO4(aq), which is yellow, is added to a colorless aqueous solution containing barium nitrate, Ba(NO3)2(aq), a yellow solid forms (see Figure 7.1). The fact that a solid forms tells us that a reaction—a chemical change—has occurred. That is, we have a situation where
What is the equation that describes this chemical change? To write the equation, we must decipher the identities of the reactants and products. The reactants have already been described: K2 CrO4 (aq ) and Ba(NO3 )2 (aq ). Is there some way in which we can predict the identities of the products? What is the yellow solid? The best way to predict the identity of this solid is to first consider what products are possible. To do this we need to know what chemical species are present in the solution that results when the reactant solutions are mixed. First, let’s think about the nature of each reactant in an aqueous solution.
|
- Home
- Chapters Part 1
- Chapter 1
- Chapter 2>
- Chapter 3>
- Chapter 4>
- Chapter 11>
- 11.1 Rutherford's Atom
- 11.2 Electromagnetic Radiation
- 11.3 Emission of Energy by Atom
- 11.4 The Energy Levels of Hydrogen
- 11.5 The Bohr Model of the Atom
- 11.6 The Wave Mechanical Model of the Atom
- 11.7 The Hydrogen Orbitals
- 11.8 The Wave Mechanical Model: Further Development
- 11.9 Electron Arrangements in the First Eighteen Atoms on the Periodic Table
- 11.10 Electron Configuration and the Periodic Table
- 11.11 Atomic Properties and the Periodic Table
- Chapter 12>
- 12.1 Types of Chemical Bonds
- 12.2 Electronegativity
- 12.3 Bond Polarity and Dipole Moments
- 12.4 Stable Electron Configurations and Charges on Ions
- 12.5 Ionic Bonding and Structures of Ionic Compounds
- 12.6 Lewis Structures
- 12.7 Lewis Structures of Molecules with Multiple Bonds
- 12.8 Molecular Structure
- 12.9 Molecular Structure: The VSEPR Model
- 12.10 Molecular Structure: Molecules with Double Bonds
- Chapter 5>
- Chapter 6>
- Chapter 7>
- Chapters Part 2