OBJECTIVE: To learn to distinguish between physical and chemical properties. To learn to distinguish between physical and chemical changes.
Properties of Matter
Physical Properties
Are characteristics of a substance as it exist that can be observed and measurable.
Two type of physical properties.
Chemical Properties
It is the ability to form new substances under given condition.
Example:
Octane( the major component of gasoline) + O2+ spark --> combustion( release of energy) + CO2 +H2O
Octane changes in to CO2 and H2O after ignition
Wood burning in a fireplace, giving off heat and gases and leaving a residue of ashes. In this process, the wood is changed to several new substances.
Identifying Physical and Chemical Properties
Classify each of the following as a physical or a chemical property.
a. The boiling point of a certain alcohol is 78 °C.
b. Diamond is very hard.
c. Sugar ferments to form alcohol.
d. A metal wire conducts an electric current.
S O L U T I O N
Items (a), (b), and (d) are physical properties; they describe inherent characteristics of each substance, and no change in composition occurs. A metal wire has the same composition before and after an electric current has passed through it. Item (c) is a chemical property of sugar. Fermentation of sugars involves the formation of a new substance (alcohol).
Physical and Chemical Change
All matter can go into Physical and chemical change.
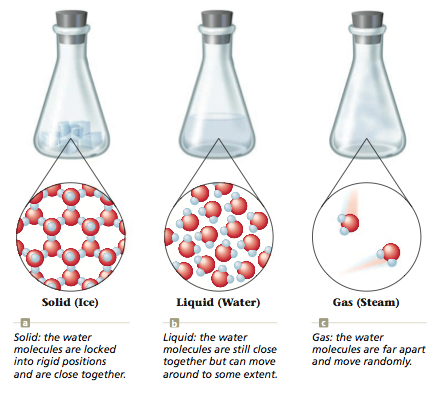
- The difference between the three states of matter is the spacing of the molecule and how ridged the spacing of the molecules.
Different States of Water
- Physical properties
- Chemical properties
Physical Properties
Are characteristics of a substance as it exist that can be observed and measurable.
Two type of physical properties.
- Intrinsic physical properties
- Extrinsic physical properties
Chemical Properties
It is the ability to form new substances under given condition.
Example:
Octane( the major component of gasoline) + O2+ spark --> combustion( release of energy) + CO2 +H2O
Octane changes in to CO2 and H2O after ignition
Wood burning in a fireplace, giving off heat and gases and leaving a residue of ashes. In this process, the wood is changed to several new substances.
Identifying Physical and Chemical Properties
Classify each of the following as a physical or a chemical property.
a. The boiling point of a certain alcohol is 78 °C.
b. Diamond is very hard.
c. Sugar ferments to form alcohol.
d. A metal wire conducts an electric current.
S O L U T I O N
Items (a), (b), and (d) are physical properties; they describe inherent characteristics of each substance, and no change in composition occurs. A metal wire has the same composition before and after an electric current has passed through it. Item (c) is a chemical property of sugar. Fermentation of sugars involves the formation of a new substance (alcohol).
Physical and Chemical Change
All matter can go into Physical and chemical change.
- Lets look at water.
- The difference between the three states of matter is the spacing of the molecule and how ridged the spacing of the molecules.
Different States of Water
This is a good example of physical change because the water molecules never change, but the physical spacing of the water molecules changes.
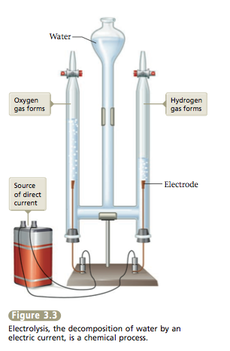
Example of Chemical Change with Water
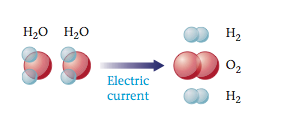
By adding electricity to water, we can break water (H2O) to Hydrogen gas( H2) and Oxygen gas( O2).
Example of Chemical Change with Water
By adding electricity to water, we can break water (H2O) to Hydrogen gas( H2) and Oxygen gas( O2).
- The name for the decomposition of water by electricity is Electrolysis.
S U M M A R I Z E
Physical and Chemical Changes
Physical and Chemical Changes
- A physical change involves a change in one or more physical properties, but no change in the fundamental components that make up the substance. The most common physical changes are changes of state: solid ⇔ liquid ⇔ gas.
- A chemical change involves a change in the fundamental components of the substance; a given substance changes into a different substance or substances. Chemical changes are called reactions: silver tarnishes by reacting with substances in the air; a plant forms a leaf by combining various substances from the air and soil; and so on.