OBJECTIVE: To understand molecular structure and bond angles.
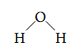
So far in this chapter we have considered the Lewis structures of molecules. These structures represent the arrangement of the valence electrons in a mol- ecule. We use the word structure in another way when we talk about the molecular structure or geometric structure of a molecule. These terms refer to the three-dimensional arrangement of the atoms in a molecule. For example, the water molecule is known to have the molecular structure
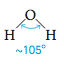
which is often called “bent” or “V-shaped.” To describe the structure more precisely, we often specify the bond angle. For the H2O molecule the bond angle is about 105°.
|
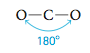
On the other hand, some molecules exhibit a linear structure (all atoms in a line). An example is the CO2 molecule.
Note that a linear molecule has a 180° bond angle.
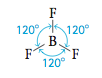
A third type of molecular structure is illustrated by BF3, which is planar or flat (all four atoms in the same plane) with 120° bond angles. The name usually given to this structure is trigonal planar structure, al- though triangular might seem to make more sense.
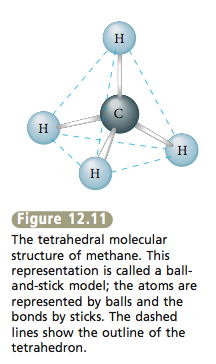
Another type of molecular structure is illustrated by methane, CH4. This molecule has the molecular structure shown in Figure 12.11, which is called a tetrahedral structure or a tetrahedron. The dashed lines shown connecting the H atoms define the four identical triangular faces of the tetrahedron. |