OBJECTIVE: To learn about Bohr’s model of the hydrogen atom.
|
In 1911 at the age of twenty-five, Niels Bohr (Figure 11.16) received his Ph.D. in physics. He was convinced that the atom could be pictured as a small positive nucleus with electrons orbiting around it.
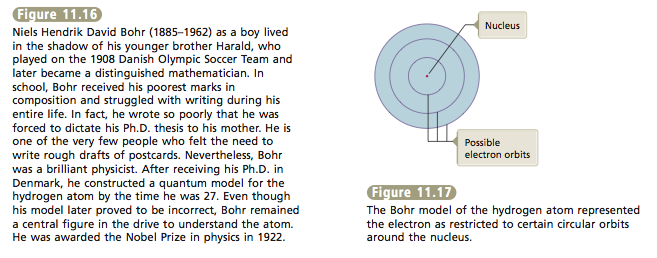
Over the next two years, Bohr constructed a model of the hydrogen atom with quantized energy levels that agreed with the hydrogen emission results we have just discussed. Bohr pictured the electron moving in circular orbits corresponding to the various allowed energy levels. He suggested that the electron could jump to a different orbit by absorbing or emitting a pho- ton of light with exactly the correct energy content. Thus, in the Bohr atom, the energy levels in the hydrogen atom represented certain allowed circular orbits (Figure 11.17). At first Bohr’s model appeared very promising. It fit the hydrogen atom very well. However, when this model was applied to atoms other than hy- drogen, it did not work. In fact, further experiments showed that the Bohr model is fundamentally incorrect. Although the Bohr model paved the way for later theories, it is important to realize that the current theory of atomic structure is not the same as the Bohr model. Electrons do not move around the nucleus in circular orbits like planets orbiting the sun. |