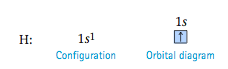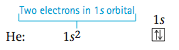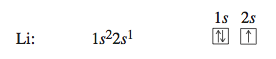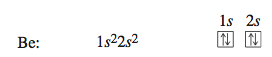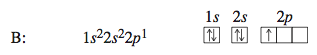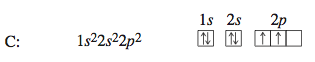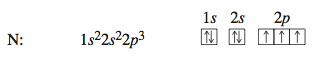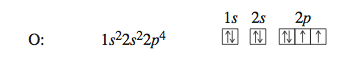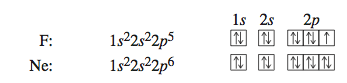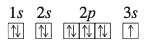OBJECTIVE: To understand how the principal energy levels fill with electrons in atoms beyond hydrogen. To learn about valence electrons and core electrons.
The most attractive orbital to an electron in an atom is always the 1s, because in this orbital the negatively charged electron is closer to the positively charged nucleus than in any other orbital. That is, the 1s orbital involves the space around the nucleus that is closest to the nucleus. As n increases, the orbital becomes larger—the electron, on average, occupies space farther from the nucleus.
|
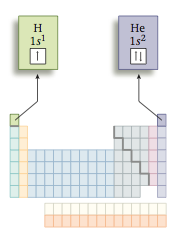
So in its ground state hydrogen has its lone electron in the 1s orbital. This is commonly represented in two ways. First, we say that hydrogen has the electron arrangement, or electron configuration, 1s1. This just means there is one electron in the 1s orbital. We can also represent this configuration by using an orbital diagram, also called a box diagram, in which orbitals are represented by boxes grouped by sublevel with small ar- rows indicating the electrons. For hydrogen, the electron configuration and box diagram are
The arrow represents an electron spinning in a particular direction. The next element is helium, Z = 2. It has two protons in its nucleus and so has two electrons. Because the 1s orbital is the most desirable, both electrons go there but with opposite spins. For helium, the electron configuration and box diagram are
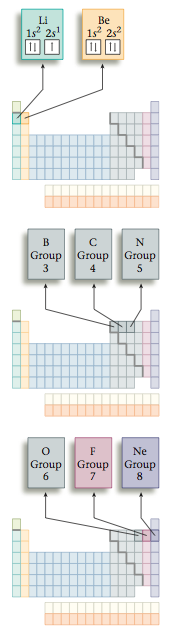
The opposite electron spins are shown by the opposing arrows in the box. Lithium (Z = 3) has three electrons, two of which go into the 1s orbital. That is, two electrons fill that orbital. The 1s orbital is the only orbital for n 1, so the third electron must occupy an orbital with n = 2—in this case the 2s orbital. This gives a 1s22s1 configuration. The electron configuration and box diagram are
The next element, beryllium, has four electrons, which occupy the 1s and 2s orbitals with opposite spins.
Boron has five electrons, four of which occupy the 1s and 2s orbitals. The fifth electron goes into the second type of orbital with n = 2, one of the 2p orbitals.
Because all the 2p orbitals have the same energy, it does not matter which 2p orbital the electron occupies.
Carbon, the next element, has six electrons: two electrons occupy the 1s orbital, two occupy the 2s orbital, and two occupy 2p orbitals. There are three 2p orbitals, so each of the mutually repulsive electrons occupies a dif- ferent 2p orbital. For reasons we will not consider, in the separate 2p orbitals the electrons have the same spin. The configuration for carbon could be written 1s^2 2s^2 2p^1 2p^1 to indicate that the electrons occupy separate 2p orbitals. However, the configuration is usually given as 1s^2 2s^2 2p^2, and it is understood that the electrons are in different 2p orbitals. Note the like spins for the unpaired electrons in the 2p orbitals.
The configuration for nitrogen, which has seven electrons, is 1s^2 2s^2 2p^3. The three electrons in 2p orbitals occupy separate orbitals and have like spins. The configuration for oxygen, which has eight electrons, is 1s^2 2s^2 2p^4. One of the 2p orbitals is now occupied by a pair of electrons with opposite spins, as required by the Pauli exclusion principle.
|
The electron configurations and orbital diagrams for fluorine (nine electrons) and neon (ten electrons) are
With neon, the orbitals with n = 1 and n = 2 are completely filled.
For sodium, which has eleven electrons, the first ten electrons occupy the 1s, 2s, and 2p orbitals, and the eleventh electron must occupy the first orbital with n = 3, the 3s orbital. The electron configuration for sodium is 1s^2 2s^2 2p^6 3s^1. To avoid writing the inner-level electrons, we often abbreviate the configuration 1s^2 2s^2 2p^6 3s^1 as [Ne]3s1, where [Ne] represents the electron configuration of neon, 1s^2 2s^2 2p^6.
The orbital diagram for sodium is
For sodium, which has eleven electrons, the first ten electrons occupy the 1s, 2s, and 2p orbitals, and the eleventh electron must occupy the first orbital with n = 3, the 3s orbital. The electron configuration for sodium is 1s^2 2s^2 2p^6 3s^1. To avoid writing the inner-level electrons, we often abbreviate the configuration 1s^2 2s^2 2p^6 3s^1 as [Ne]3s1, where [Ne] represents the electron configuration of neon, 1s^2 2s^2 2p^6.
The orbital diagram for sodium is
The next element, magnesium, Z = 12, has the electron configuration 1s^2 2s^2 2p^6 3s^2, or [Ne]3s2.
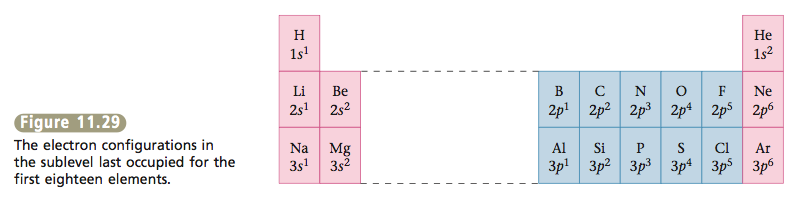
The next six elements, aluminum through argon, have electron config- urations obtained by filling the 3p orbitals one electron at a time. Figure 11.29 summarizes the electron configurations of the first eighteen elements by giving the number of electrons in the type of orbital (sublevel) occupied last.
The next six elements, aluminum through argon, have electron config- urations obtained by filling the 3p orbitals one electron at a time. Figure 11.29 summarizes the electron configurations of the first eighteen elements by giving the number of electrons in the type of orbital (sublevel) occupied last.
At this point it is useful to introduce the concept of valence electrons—that is, the electrons in the outermost (highest) principal energy level of an atom. For example, nitrogen, which has the electron configuration 1s^2 2s^2 2p^3, has electrons in principal levels 1 and 2. Therefore, level 2 (which has 2s and 2p sublevels) is the valence level of nitrogen, and the 2s and 2p electrons are the valence electrons. For the sodium atom (electron configuration 1s^2 2s^2 2p^6 3s^1, or [Ne]3s1), the valence electron is the electron in the 3s orbital, because in this case principal energy level 3 is the outermost level that contains an electron. The valence electrons are the most important elec- trons to chemists because, being the outermost electrons, they are the ones involved when atoms attach to each other (form bonds), as we will see in the next chapter. The inner electrons, which are known as core electrons, are not involved in bonding atoms to each other.
Note in Figure 11.29 that a very important pattern is developing: except for helium, the atoms of elements in the same group (vertical column of the peri- odic table) have the same number of electrons in a given type of orbital (sublevel), except that the orbitals are in different principal energy levels. Remember that the elements were originally organized into groups on the periodic table on the basis of similarities in chemical properties. Now we understand the reason behind these groupings. Elements with the same valence electron arrangement show very similar chemical behavior.