OBJECTIVE: To learn to distinguish between mixtures and pure substances.
Virtually all of the matter around us consists of mixtures of substances. For example, if you closely observe a sample of soil, you will see that it has many types of components, including tiny grains of sand and remnants of plants. The air we breathe is a complex mixture of such gases as oxygen, nitrogen, carbon dioxide, and water vapor. Even the sparkling water from a drinking fountain contains many substances besides water.
A mixture can be defined as something that has variable composition.
A pure substance, on the other hand, will always have the same composition. Pure substances are either elements or compounds.
A mixture can be defined as something that has variable composition.
- For example, wood is a mixture (its composition varies greatly depending on the tree from which it originates); wine is a mixture (it can be red or pale yellow, sweet or dry); coffee is a mixture (it can be strong, weak, or bitter); and, although it looks very pure, water pumped from deep in the earth is a mixture (it contains dissolved minerals and gases).
A pure substance, on the other hand, will always have the same composition. Pure substances are either elements or compounds.
- For example, pure water is a compound containing individual H2O molecules. How- ever, as we find it in nature, liquid water always contains other substances in addition to pure water—it is a mixture.
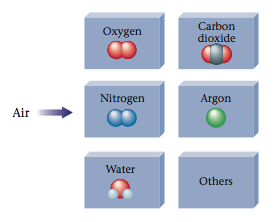
- For example, the mixture known as air can be separated into oxygen (ele- ment), nitrogen (element), water (compound), carbon dioxide (compound), argon (element), and other pure substances.
Mixtures can be classified as either homogeneous or heterogeneous.
Distinguishing Between Mixtures and Pure Substances
Identify each of the following as a pure substance, a homogeneous mixture, or a heterogeneous mixture.
a. gasoline
b. a stream with gravel at the bottom
c. air
d. brass
e. copper metal
S O L U T I O N
a. Gasoline is a homogeneous mixture containing many compounds.
b. A stream with gravel on the bottom is a heterogeneous mixture.
c. Air is a homogeneous mixture of elements and compounds.
d. Brass is a homogeneous mixture containing the elements copper and zinc. Brass is not a pure substance because the relative amounts of copper and zinc are different in different brass samples.
e. Copper metal is a pure substance (an element).
Extra Link
You can also visit this link. It could help improve your understanding on mixtures and pure substances.
- Homogeneous mixture is the same proportion throughout the material.
- Heterogenous mixture contains regions of different proportion.
Distinguishing Between Mixtures and Pure Substances
Identify each of the following as a pure substance, a homogeneous mixture, or a heterogeneous mixture.
a. gasoline
b. a stream with gravel at the bottom
c. air
d. brass
e. copper metal
S O L U T I O N
a. Gasoline is a homogeneous mixture containing many compounds.
b. A stream with gravel on the bottom is a heterogeneous mixture.
c. Air is a homogeneous mixture of elements and compounds.
d. Brass is a homogeneous mixture containing the elements copper and zinc. Brass is not a pure substance because the relative amounts of copper and zinc are different in different brass samples.
e. Copper metal is a pure substance (an element).
Extra Link
You can also visit this link. It could help improve your understanding on mixtures and pure substances.