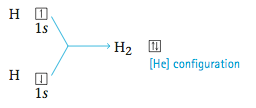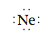OBJECTIVE: To learn to write Lewis structures.
|
Bonding involves just the valence electrons of atoms. Valence electrons are transferred when a metal and a nonmetal react to form an ionic compound. Valence electrons are shared between nonmetals in covalent bonds.
The Lewis structure is a representation of a molecule that shows how the valence electrons are arranged among the atoms in the molecule. These representations are named after G. N. Lewis, who conceived the idea while lecturing to a class of general chemistry students in 1902. The rules for writing Lewis structures are based on observations of many molecules from which chemists have learned that the most important requirement for the formation of a stable compound is that the atoms achieve noble gas electron configurations. No dots are shown on the K+ ion because it has lost its only valence electron (the 4s electron). The Br- ion is shown with eight electrons because it has a filled valence shell.
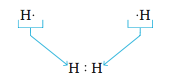
Next we will consider Lewis structures for molecules with covalent bonds, involving nonmetals in the first and second periods. The principle of achieving a noble gas electron configuration applies to these elements as follows: 1. Hydrogen forms stable molecules where it shares two electrons. That is, it follows a duet rule. For example, when two hydrogen atoms, each with one electron, combine to form the H2 molecule, we have By sharing electrons, each hydrogen in H2 has, in effect, two electrons; that is, each hydrogen has a filled valence shell.
2. Helium does not form bonds because its valence orbital is already filled; it is a noble gas. Helium has the electron configuration 1s2 and can be represented by the Lewis structure
|
|
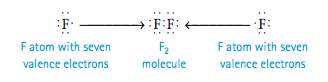
3. The second-row nonmetals carbon through fluorine form stable molecules when they are surrounded by enough electrons to fill the valence orbitals—that is, the one 2s and the three 2p orbitals. Eight electrons are required to fill these orbitals, so these elements typically obey the octet rule; they are surrounded by eight electrons. An example is the F2 molecule, which has the following Lewis structure:
Note that only the valence electrons (2s22p6) of the neon atom are represented by the Lewis structure. The 1s2 electrons are core electrons and are not shown.
|
|
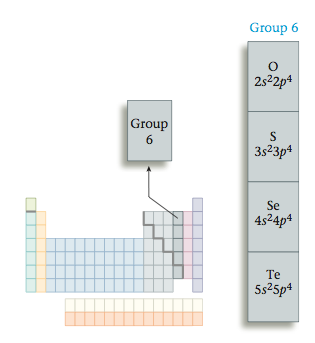
Next we want to develop some general procedures for writing Lewis structures for molecules. Remember that Lewis structures involve only the valence electrons of atoms, so before we proceed, we will review the relationship of an element’s position on the periodic table to the number of valence electrons it has. Recall that the group number gives the total number of valence electrons. For example, all Group 6 elements have six valence electrons (valence configuration ns2np4).
|
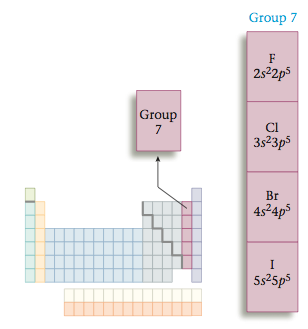
Similarly, all Group 7 elements have seven valence electrons (valence configuration ns2np5).
In writing the Lewis structure for a molecule, we need to keep the following things in mind:
- We must include all the valence electrons from all atoms. The total number of electrons available is the sum of all the valence electrons from all the atoms in the molecule.
- Atoms that are bonded to each other share one or more pairs of electrons.
- The electrons are arranged so that each atom is surrounded by enough electrons to fill the valence orbitals of that atom. This means two electrons for hydrogen and eight electrons for second- row nonmetals.