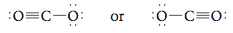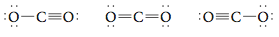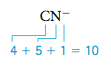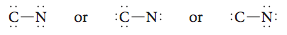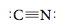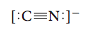OBJECTIVE: To learn how to write Lewis structures for molecules with multiple bonds.
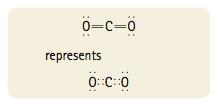
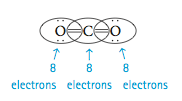
Now each atom is surrounded by eight electrons, and the total number of electrons is sixteen, as required. This is the correct Lewis structure for carbon dioxide, which has two double bonds. A single bond involves two atoms sharing one electron pair. A double bond involves two atoms shar- ing two pairs of electrons.
In considering the Lewis structure for CO2, you may have come up with Note that both of these structures have the required sixteen electrons and that both have octets of electrons around each atom (verify this for yourself). Both of these structures have a triple bond in which three electron pairs are shared. Are these valid Lewis structures for CO2? Yes. So there really are three Lewis structures for CO2:
This brings us to a new term, resonance. A molecule shows resonance when more than one Lewis structure can be drawn for the molecule. In such a case we call the various Lewis structures resonance structures.
Of the three resonance structures for CO2 shown above, the one in the center with two double bonds most closely fits our experimental information about the CO2 molecule. In this text we will not be concerned about how to choose which resonance structure for a molecule gives the “best” description of that molecule’s properties. Next let’s consider the Lewis structure of the CN- (cyanide) ion. Step 1 Summing the valence electrons, we have
Note that the negative charge means an extra electron must be added.
Step 2 Draw a single bond (CON). Step 3 Next, we distribute the remaining electrons to achieve a noble gas configuration for each atom. Eight electrons remain to be distributed. We can try various possibilities, such as These structures are incorrect. To show why none is a valid Lewis structure, count the electrons around the C and N atoms. In the left structure, neither atom satisfies the octet rule. In the center structure, C has eight electrons but N has only four. In the right structure, the opposite is true. Remember that both atoms must simultaneously satisfy the octet rule. Therefore, the correct arrangement is
|
|
(Satisfy yourself that both carbon and nitrogen have eight electrons.) In this case we have a triple bond between C and N, in which three electron pairs are shared. Because this is an anion, we indicate the charge outside of square brackets around the Lewis structure.
In summary, sometimes we need double or triple bonds to satisfy the octet rule. Writing Lewis structures is a trial-and-error process. Start with single bonds between the bonded atoms and add multiple bonds as needed.
We will write the Lewis structure for NO2- in Example 12.3 to make sure the procedures for writing Lewis structures are clear. |