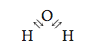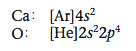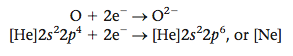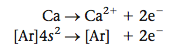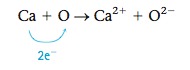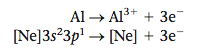OBJECTIVE: To learn about stable electron configurations. To learn to predict the formulas of ionic compounds.
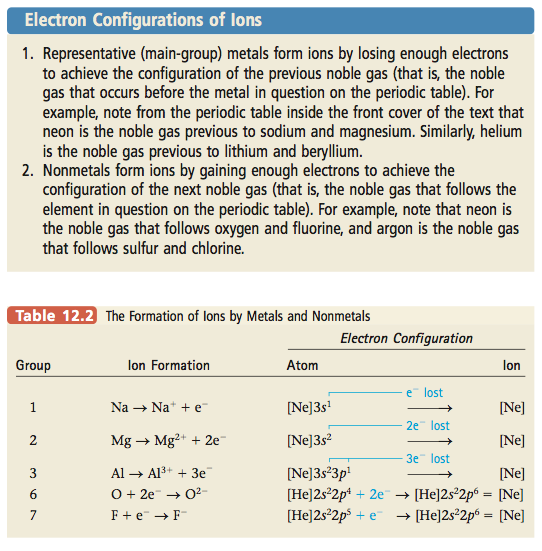
Notice something very interesting about the ions in Table 12.2: they all have the electron configuration of neon, a noble gas. That is, sodium loses its one valence electron (the 3s) to form Na+, which has an [Ne] electron configuration. Likewise, Mg loses its two valence electrons to form Mg2+, which also has an [Ne] electron configuration. On the other hand, the nonmetal atoms gain just the number of electrons needed for them to achieve the noble gas electron configuration. The O atom gains two electrons and the F atom gains one electron to give O2- and F-, respectively, both of which have the [Ne] electron configuration. We can summarize these observations as follows:
|
This brings us to an important general principle. In observing mil- lions of stable compounds, chemists have learned that in almost all stable chemical compounds of the representative elements, all of the atoms have achieved a noble gas electron configuration. The importance of this observation cannot be overstated. It forms the basis for all of our fundamental ideas about why and how atoms bond to each other.
We have already seen this principle operating in the formation of ions (see Table 12.2). We can summarize this behavior as follows: when representative metals and nonmetals react, they transfer electrons in such a way that both the cation and the anion have noble gas electron configurations. On the other hand, when nonmetals react with each other, they share electrons in ways that lead to a noble gas electron configuration for each atom in the resulting molecule. For example, oxygen ([He]2s22p4), which needs two more electrons to achieve an [Ne] configuration, can get these electrons by combining with two H atoms (each of which has one electron), to form water, H2O. This fills the valence orbitals of oxygen. In addition, each H shares two electrons with the oxygen atom,
which fills the H 1s orbital, giving it a 1s2 or [He] electron configuration. We will have much more to say about covalent bonding in Section 12.6.
At this point let’s summarize the ideas we have introduced so far. |
|
To show how to predict what ions form when a metal reacts with a non- metal, we will consider the formation of an ionic compound from calcium and oxygen. We can predict what compound will form by considering the valence electron configurations of the following two atoms:
From Figure 12.3 we see that the electronegativity of oxygen (3.5) is much greater than that of calcium (1.0), giving a difference of 2.5. Because of this large difference, electrons are transferred from calcium to oxygen to form an oxygen anion and a calcium cation. How many electrons are trans- ferred? We can base our prediction on the observation that noble gas configurations are the most stable. Note that oxygen needs two electrons to fill its valence orbitals (2s and 2p) and achieve the configuration of neon (1s22s22p6), which is the next noble gas.
And by losing two electrons, calcium can achieve the configuration of argon (the previous noble gas).
Two electrons are therefore transferred as follows:
To predict the formula of the ionic compound, we use the fact that chemical compounds are always electrically neutral—they have the same total quantities of positive and negative charges. In this case we must have equal numbers of Ca2+ and O2- ions, and the empirical formula of the compound is CaO.
The same principles can be applied to many other cases. For example, consider the compound formed from aluminum and oxygen. Aluminum has the electron configuration [Ne]3s23p1. To achieve the neon configuration, aluminum must lose three electrons, forming the Al3+ ion. |
|
Therefore, the ions will be Al3+ and O2-. Because the compound must be electrically neutral, there will be three O2- ions for every two Al3+ ions, and the compound has the empirical formula Al2O3.
Table 12.3 shows common elements that form ions with noble gas electron configurations in ionic compounds. Notice that our discussion in this section refers to metals in Groups 1, 2, and 3 (the representative metals). The transition metals exhibit more complicated behavior (they form a variety of ions), which we will not be concerned with in this text. |