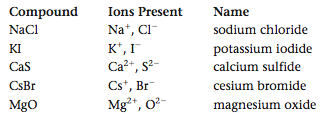OBJECTIVE: To learn to name binary compounds of a metal and a nonmetal.
|
As we saw in Section 4.11, when a metal such as sodium combines with a nonmetal such as chlorine, the resulting compound contains ions. The metal loses one or more electrons to become a cation, and the nonmetal gains one or more electrons to form an anion. The resulting substance is called a binary ionic compound. Binary ionic compounds contain a positive ion (cation), which is always written first in the formula, and a negative ion (anion). To name these compounds we simply name the ions.
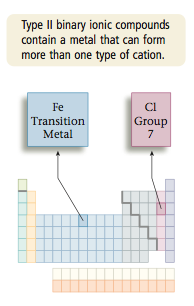
Other metal atoms can form two or more cations. For example, Cr can form Cr2+ and Cr3+ and Cu can form Cu+ and Cu2+. We will call such ions Type II cations and their compounds Type II binary compounds.
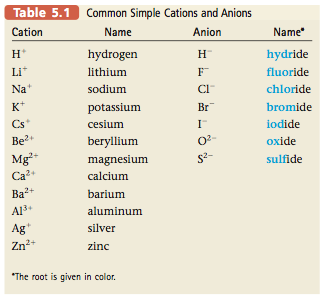
In summary: Type I compounds: The metal present forms only one type of cation. Type II compounds: The metal present can form two (or more) cations that have different charges. Some common cations and anions and their names are listed in Table 5.1. You should memorize these. They are an essential part of your chemical vocabulary. |
For example, the compound NaI is called sodium iodide. It contains Na+ (the sodium cation, named for the parent metal) and I- (iodide: the root of iodine plus -ide). Similarly, the compound CaO is called calcium oxide because it contains Ca2+ (the calcium cation) and O2- (the oxide anion).
The rules for naming binary compounds are also illustrated by the following examples:
The rules for naming binary compounds are also illustrated by the following examples:
|
As we said in the previous section, we can predict with certainty that each Group 1 metal will give a 1+ cation and each Group 2 metal will give a 2+ cation. Aluminum always forms Al3+.
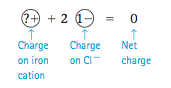
However, there are many metals that can form more than one type of cation. For example, lead (Pb) can form Pb2+ or Pb4+ in ionic compounds. Also, iron (Fe) can produce Fe2+ or Fe3+, chromium (Cr) can produce Cr2+ or Cr3+, gold (Au) can produce Au+ or Au3+, and so on. This means that if we saw the name gold chloride, we wouldn’t know whether it referred to the compound AuCl (containing Au and Cl) or the compound AuCl3 (con- taining Au3+ and three Cl- ions). Therefore, we need a way of specifying which cation is present in compounds containing metals that can form more than one type of cation. Chemists have decided to deal with this situation by using a Roman numeral to specify the charge on the cation. To see how this works, consider the compound FeCl2. Iron can form Fe2+ or Fe3+, so we must first decide which of these cations is present. We can determine the charge on the iron cation, because we know it must just balance the charge on the two 1- an- ions (the chloride ions). Thus if we represent the charges as |
The compound FeCl2, then, contains one Fe2+ ion and two Cl- ions. We call this compound iron(II) chloride, where the II tells the charge of the iron cation. That is, Fe2+ is called iron(II). Likewise, Fe3+ is called iron(III). And FeCl3, which contains one Fe3+ ion and three Cl- ions, is called iron(III) chloride. Remember that the Roman numeral tells the charge on the ion, not the number of ions present in the compound.
To help distinguish between Type I and Type II cations, remember that Group 1 and 2 metals are always Type I. On the other hand, transition metals are almost always Type II.
Games
Click here to play a game!