OBJECTIVE: To learn about Dalton’s theory of atoms. To understand and illustrate the law of constant composition.
|
As scientists of the eighteenth century studied the nature of materials, several things became clear:
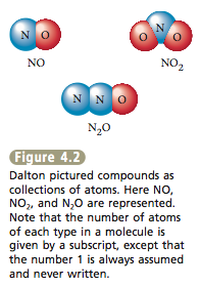
1. Most natural materials are mixtures of pure substances. 2. Pure substances are either elements or combinations of elements called compounds. 3. A given compound always contains the same proportions (by mass) of the elements. For example, water always contains 8 g of oxygen for every 1 g of hydrogen, and carbon dioxide always contains 2.7 g of oxygen for every 1 g of carbon. This principle became known as the law of constant composition. It means that a given compound always has the same composition, regardless of where it comes from. Dalton’s model successfully explained important observations such as the law of constant composition. This law makes sense because if a compound always contains the same relative numbers of atoms, it will always contain the same proportions by mass of the various elements.
|