OBJECTIVE: To explore the nature of electromagnetic radiation.
Energy is being transmitted from one place to another by light—more properly called electromagnetic radiation. Many kinds of electromagnetic radiation exist, including the X rays used to make images of bones, the “white” light from a light bulb, the microwaves used to cook hot dogs and other food, and the radio waves that transmit voices and music.
|
A particular wave is characterized by three properties: wavelength, frequency, and speed.
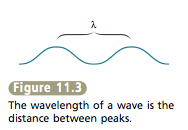
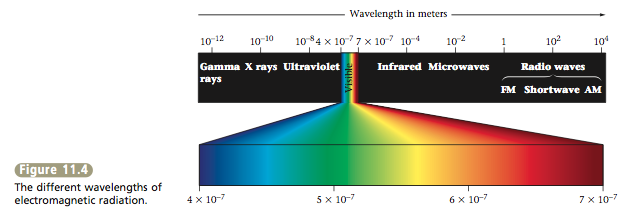
The wavelength (symbolized by the Greek letter lambda, λ) is the distance between two consecutive wave peaks (see Figure 11.3). The frequency of the wave (symbolized by the Greek letter nu, v) indicates how many wave peaks pass a certain point per given time period. The various types of electromagnetic radiation (X rays, microwaves, and so on) differ in their wavelengths. The classes of electromagnetic radiation are shown in Figure 11.4. Notice that X rays have very short wavelengths, whereas radiowaves have very long wavelengths. |
|
Radiation provides an important means of energy transfer. Visualize electromagnetic radiation (“light”) as a wave that carries energy through space. Sometimes, however, light doesn’t behave as though it were a wave. That is, electromagnetic radiation can sometimes have properties that are characteristic of particles. (You will learn more about this idea in later courses.) Another way to think of a beam of light traveling through space, then, is as a stream of tiny packets of energy called photons.
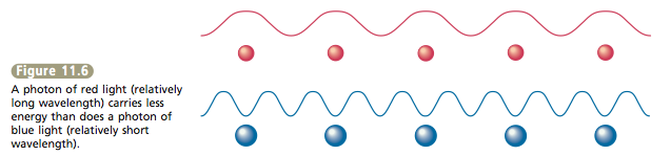
What is the exact nature of light? Does it consist of waves or is it a stream of particles of energy? It seems to be both (see Figure 11.5). This situ- ation is often referred to as the wave–particle nature of light. Different wavelengths of electromagnetic radiation carry different amounts of energy. For example, the photons that correspond to red light carry less energy than the photons that correspond to blue light. In general, the longer the wavelength of light, the lower the energy of its photons (see Figure 11.6). |
Speed of light is 2.99*10^(8)m/s through space.
- A particular wave is characterized by three properties.
- Wavelength
- Frequency
- Speed
- A particular wave is characterized by three properties.
- Wavelength (λ)
- Is the distance between 2 consecutive peaks or 2 consecutive valleys
- Frequency (F)
- -indicates how many waves peaks in a given time.
- Speed( v)
- - is how fast the wave is moving.
- Wavelength Si unit is the same as distance( m,mm,nm)
- Unit of Frequency is Hz
- Si unit for time?
Equation
- s= λ*f
- Speed=wavelength*frequency
Example problem 1.
- If the wavelength is 10 m and the frequency is 2/s, what is the wave speed.
- υ=λf
- υ= 10m*2/s
- υ=20m/s