When dissolved in water, certain molecules produce H+ ions (protons). These substances, which are called acids, were first recognized by the sour taste of their solutions. For example, citric acid is responsible for the tartness of lemons and limes. Acids will be discussed in detail later. Here we simply present the rules for naming acids.
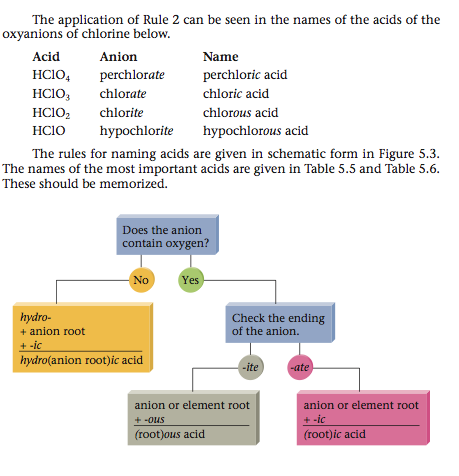
An acid can be viewed as a molecule with one or more H+ ions attached to an anion. The rules for naming acids depend on whether the anion contains oxygen.
An acid can be viewed as a molecule with one or more H+ ions attached to an anion. The rules for naming acids depend on whether the anion contains oxygen.
Extra Link
Click here
Games
Click here for a jeopardy review game!