OBJECTIVE: To understand the nature of bonds and their relationship to electronegativity.
Two identical atoms react to form a co- valent bond in which electrons are shared equally. When different nonmetals react, a bond forms in which electrons are shared unequally, giving a polar covalent bond. The unequal sharing of electrons between two atoms is described by a property called electronegativity: the relative ability of an atom in a molecule to attract shared electrons to itself.
|
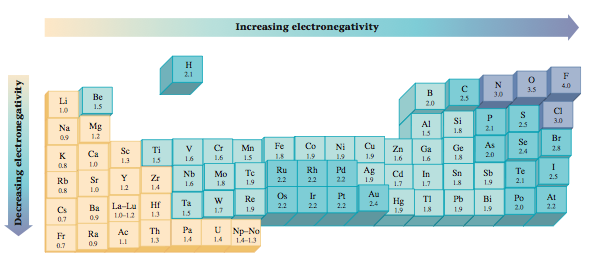
Chemists determine electronegativity values for the elements (Figure 12.3) by measuring the polarities of the bonds between various atoms. Note that electronegativity generally increases going from left to right across a period and decreases going down a group for the representative elements. The range of electronegativity values is from 4.0 for fluorine to 0.7 for cesium and francium. Remember, the higher the atom’s electronegativity value, the closer the shared electrons tend to be to that atom when it forms a bond.
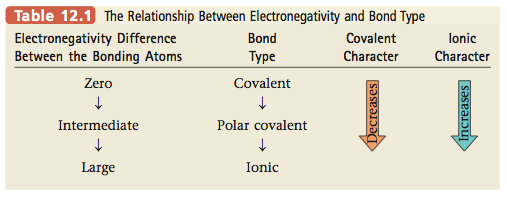
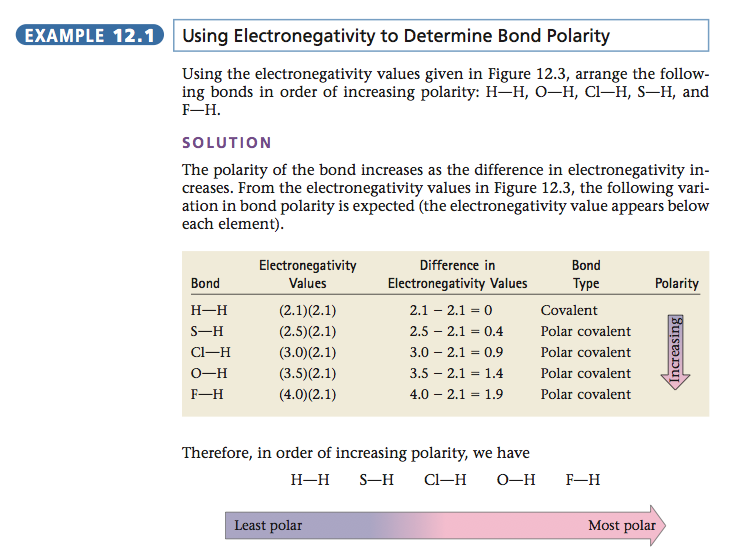
The polarity of a bond depends on the difference between the electronegativity values of the atoms forming the bond. If the atoms have very similar electronegativities, the electrons are shared almost equally and the bond shows little polarity. If the atoms have very different electronegativity values, a very polar bond is formed. In extreme cases one or more electrons are actually transferred, forming ions and an ionic bond. For example, when an element from Group 1 (electronegativity values of about 0.8) reacts with an element from Group 7 (electronegativity values of about 3), ions are formed and an ionic substance results. The relationship between electronegativity and bond type is shown in Table 12.1. The various types of bonds are summarized in Figure 12.4. |
Video Link
Click here for a video explaining electronegativity.
Extra Link
Click here for an extra link.
Games
Click here.