OBJECTIVE: To understand bond polarity and how it is related to molecular polarity.
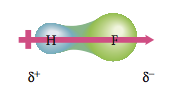
We saw in Section 12.1 that hydrogen fluoride has a positive end and a negative end. A molecule such as HF that has a center of positive charge and a center of negative charge is said to have a dipole moment. The dipolar character of a molecule is often represented by an arrow. This arrow points toward the negative charge center, and its tail indicates the positive center of charge:
|
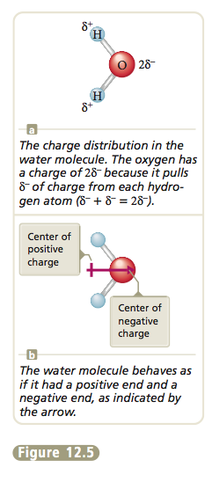
Any diatomic (two-atom) molecule that has a polar bond has a dipole moment. Some polyatomic (more than two atoms) molecules also have dipole moments. For example, because the oxygen atom in the water molecule has a greater electronegativity than the hydrogen atoms, the electrons are not shared equally. This results in a charge distribution (Figure 12.5) that causes the molecule to behave as though it had two centers of charge—one positive and one negative. So the water molecule has a dipole moment.
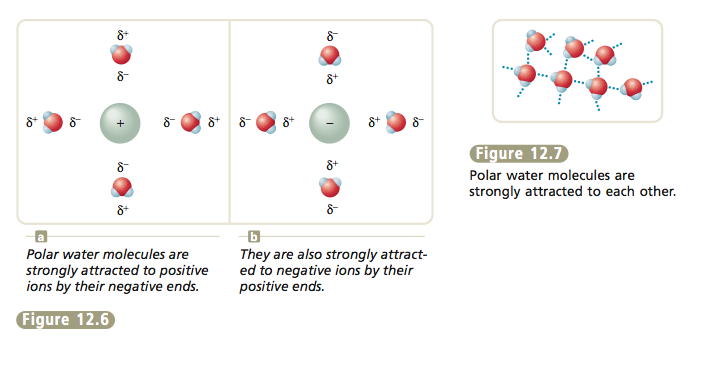
The fact that the water molecule is polar (has a dipole moment) has a profound impact on its properties. In fact, it is not overly dramatic to state that the polarity of the water molecule is crucial to life as we know it on earth. Because water molecules are polar, they can surround and attract both positive and negative ions (Figure 12.6). These attractions allow ionic materials to dissolve in water. Also, the polarity of water molecules causes them to attract each other strongly (Figure 12.7). This means that much energy is required to change water from a liquid to a gas (the molecules must be separated from each other to undergo this change of state). Therefore, it is the polarity of the water molecule that causes water to remain a liquid at the temperatures on the earth’s surface. If it were nonpolar, water would be a gas and the oceans would be empty. |